Case Report
Nov 2021, 30:4
First online: 28 December 2021
Case Report
Case Report: Stemi With Covid-19
Herawati Isnanijah SpJP (K) FIHA,1 Chyntia Monica,2 Indah Trisnawaty Sp.THT-KL,3 Yahya Berkahanto Juwana SpJP (K) PhD, FIHA, FAPSIC, FAsCC,4 Doni Firman SpJP (K) FIHA5
2Cardiology Department, Pasar Rebo General Hospital, Jakarta, Indonesia, chyntiamonica@hotmail.com
3Ear, Nose and Throat Department, Pasar Rebo General Hospital, Jakarta, Indonesia, indah.trisnawaty.ent@gmail.com
4Cardiology Department, Pasar Rebo General Hospital, Jakarta, Indonesia, yahya.juwana@gmail.com
5Department of Cardiology and Vascular Medicine, National Cardiovascular Center Harapan Kita, Jakarta, Indonesia, dr.donifirman@gmail.com
ABSTRACT
BACKGROUND
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV‑2)
causing coronavirus disease 2019 (COVID-19) has reached pandemic levels by March 2020. Patients with cardiovascular disease, particularly with cardiac injury represent a vulnerable population and increased risk of mortality and morbidity. There is still no guidelines for management of cardiovascular disease during the COVID-19 pandemic.
CASE ILLUSTRATION
An unconscious 52-year-old male brought to ER with complaints of abdominal discomfort and nausea. The patient had a cardiac arrest in ER and ROSC was obtained. The patient was intubated ECG showed anterior STEMI and primary PCI was performed. The endotracheal tube was changed due to blockage of excessive and thick slime. Tracheostomy was performed. Bronchoscopy was performed and found tracheal mucosal edema, hyperemic and easily bleed; mucous plug and blood clots in the tip of tracheostomy cannula.
Thoracic CT-Scan showed ground-glass appearance and fibrosis of the 6th thoracic dextra segment. PCR SARS-CoV-2 showed reactive. The patient was discharged from our hospital after three weeks with clinically stable and referred to COVID-19 center hospital nearby his home for another two weeks. After PCR SARS CoV-2 was performed twice showed negative results, the patient was discharged.
CONCLUSION
SARS-CoV‑2 infection may lead to acute myocardial injury
through viral systemic inflammation, although specific mechanism remained uncertain. A thick mucus plaque and stool cell may be a specific clinical features in COVID-19 patients. Tracheostomy has a continuing role in managing weaning from extended periods of mechanical ventilation during the COVID-19 pandemic.
BACKGROUND
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV‑2) causing coronavirus disease 2019 (COVID-19) has reached pandemic levels by March 2020. Patients with cardiovascular disease, particularly with cardiac injury represent a vulnerable population and increased risk of mortality and morbidity.1 There is still no guidelines for management of cardiovascular disease during the COVID-19 pandemic.
CASE ILLUSTRATION
An unconscious 52-year-old male brought to ER with complaints of abdominal discomfort and nausea. The patient had a cardiac arrest in ER. After 20 cycles of CPR and four times cardioversion due to ventricular fibrillation (Illustration 1A), return of spontaneous circulation (ROSC) was obtained and the patient was intubated. The ECG showed anterior STEMI (Illustration 1B) and immediately he underwent primary percutaneous coronary intervention (PCI). During the procedure, he had four times of ventricular tachycardia (VT) and cardioversion was performed.
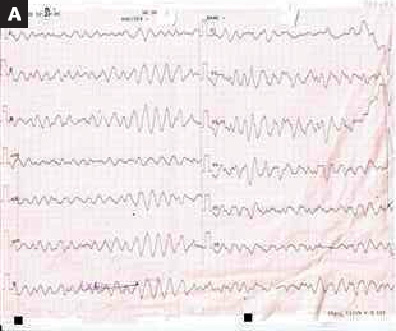
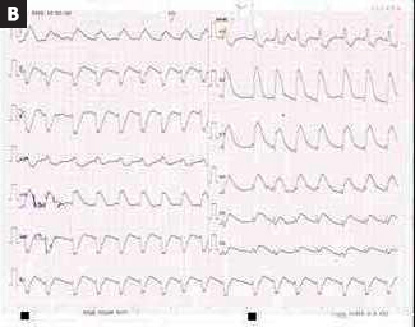
Illustration 1:
Electrocardiographic (ECG) Findings
A. ECG showed ventricular fibrillation during CPR.
B. ECG showed anterior STEMI after ROSC was obtained.
The coronary angiogram (CAG) revealed no significant stenosis in RCA and LCx, but total occlusion in proximal LAD. One drug eluting stent (DES) was placed with TIMI flow 2 result (Illustration 2).
The patient received eptifibatide and fondaparinux after the procedure besides other standard protocols medication. Echocardiography after PCI procedure showed ejection fraction 40%, hypokinesia in anterior, anteroseptal and apical wall and hyperkinesia in posterior wall.
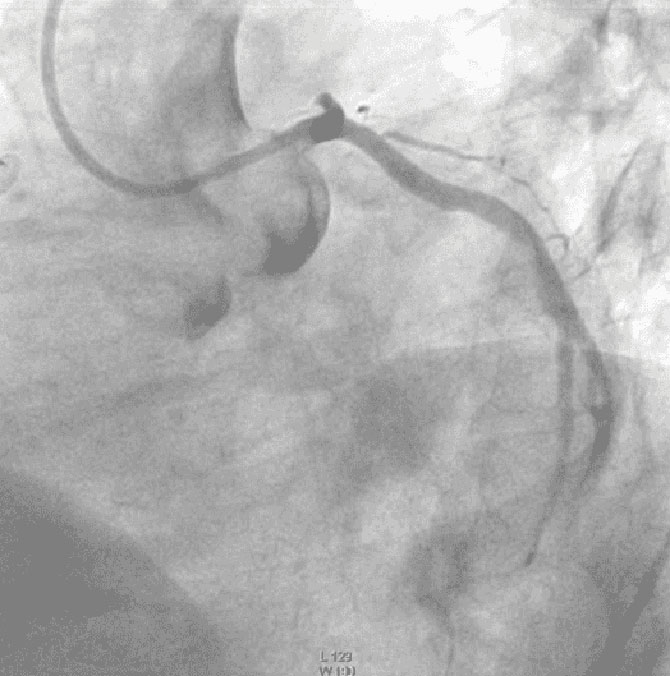
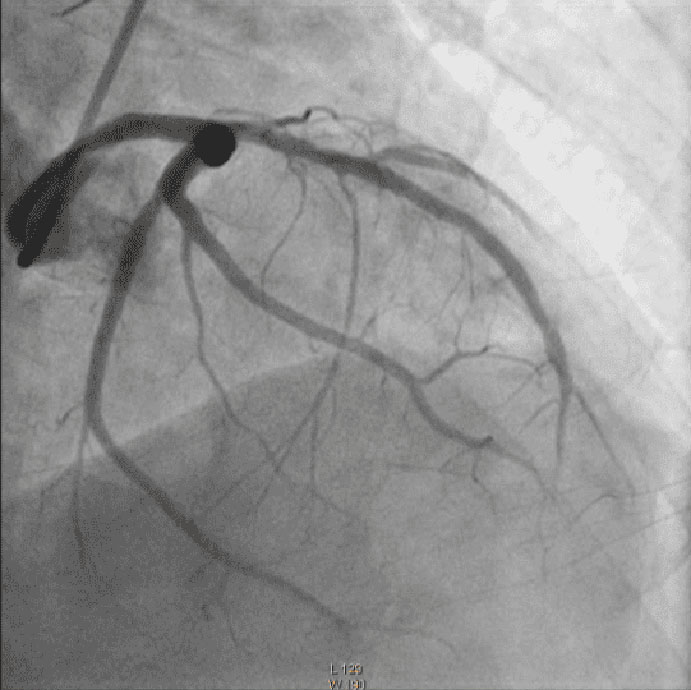
Illustration 2:
CAG showed total occlusion in proximal LAD
and TIMI flow 2 result in LAD after stent implantation.
The patient was a heavy smoker, had history of hypertension but no history of fever, cough, nor contact with COVID-19 patient but five days prior admission, the patient was travelling from Bandung to Jakarta with public transportation.
During in intensive care unit, the laboratory test showed metabolic acidosis and an increase of liver enzymes, leukocytosis, thrombocytosis, and lymphopenia.
Despite the patient was supported by ventilator the saturation remained low. The production of slime was excessive and thick resulted clot in the endotracheal tube. On the 3rd day the endotracheal tube was changed due to blockage of excessive and thick slime. On the 4th day, a recurrent event happened, during the bronchial toilet, an airway bagging was hard due to lots of stool cell and thick slime.
On the 5th day, tracheostomy was performed in patient with unknown COVID-19 status. The indication to performed tracheostomy in this patient was threatened airway obstruction due to active bleeding in trachea mixing thick jelly secretion makes mucous plugs and blood clots. Tracheostomy was performed in isolation room in CVCU with closed door during the procedure, under general anesthesia, limited staff (ENT surgeon, anesthesiologist, two nurses) wear level 3 PPE and using cuffed non-fenestrated tubes. Six hours after tracheostomy, tracheostomy cuff deflate in half. Tracheostomy care with periodic suctioning and bronchial wash which is aerosol-generating procedure, was done to overcome repeated desaturation due to mucous plug. Tracheostomy cannula do not use heat and moisture exchanger (HME) filter.
On the 7th day, bronchoscopy was performed and found tracheal mucosal edema, hyperemic and easily bleed; mucous plug and blood clots in the tip of tracheostomy cannula and cover three quarters of the tracheal diameter. Suctioning and bronchial wash performed to lift up mucous plug.
Thoracic CT-scan showed ground-glass appearance of infiltrate and fibrosis of the 6th thoracic dextra segment (Illustration 3).
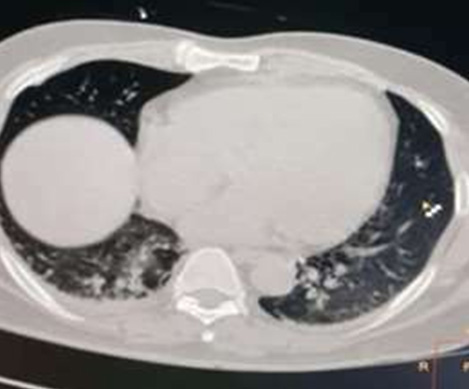
Illustration 3:
Thoracic CT-scan showed ground-glass
appearance.
PCR SARS-CoV-2 showed positive result. The patient received aseptamivir at a dose of 150 mg, levofloxacin at a dose of 500 mg, chloroquine at a dose of 1000 mg and vitamin C as treatment.
The patient was discharged from our hospital after three weeks with clinically stable and referred to COVID-19 center hospital nearby his home for another two weeks. After PCR SARS-CoV-2 was performed twice showed negative results, the patient was discharged. Recently published COVID-19 studies have shown that SARS-CoV-2 infection was more likely to occur and seem to have a heightened vulnerability to develop COVID-19 and tend to have more severe disease with worse clinical outcomes in patient with underlying cardiovascular disease (CVD).2, 3
However, there is no case report that clearly suggests that COVID-19 can cause Acute Coronary Syndrome (ACS). Patient with artery coronary disease at risk the occurrence of coronary plaque rupture secondary to virally induced systemic inflammation. The mechanism of acute myocardial injury caused by COVID-19 might be related to angiotensin converting enzyme 2 (ACE2). The interaction between SARS-CoV2 and ACE2 can result in changes of ACE2 pathways leading to acute injury to the heart, lung and endothelial cells.4, 5
In this case, theoretically, COVID-19 may induced STEMI due to systemic inflammation but we need further study to confirm direct relation between STEMI and COVID-19. The patient was admitted in our hospital 2 weeks from the first confirmed COVID-19 in Indonesia. The awareness of the sign and symptoms and logistic diagnostic tools was limited at the time but the clinical feature of this patient, massive and thick mucus productivity suspect the diagnosis of COVID-19.
The pathology of COVID-19 also shows fibromyxoid exudation and the formation of thick “mucus plugs”. A large amount of sticky secretions spilling out of the alveoli, with fibrous cords visible was found in the first autopsy.5 This case report emphasize that a thick mucus plaque with stool cell may be a specific clinical features in COVID-19 patients.
Tracheostomy is an aerosol-generating procedure, so health-care workers are at risk of infection during insertion and subsequent care, even when appropriate personal protective equipment (PPE) is used. Ensuring adequate PPE, to maintain the safety of the health-care team, is among the foremost considerations in decisions around the management of patients with COVID-19 in the ICU.
The major indication for tracheostomy remains the facilitation of mechanical ventilation for a long period, while minimizing complications from a translaryngeal endotracheal tube and weaning from ventilation. Tracheostomy might also be required for actual or threatened airway obstruction, laryngeal edema (which might be an emerging feature of COVID-19) or unsuccessful extubation due to weakness, poor cough, tenacious secretions, or a combination of these factors. Because tracheostomy is an aerosol-generating procedure and recognizing that many health-care facilities lack negative pressure rooms, portable high-efficiency particulate air filtration systems might be an acceptable alternative.
Tracheostomy to facilitate an extended period of ventilation support or weaning from ventilation initially requires a closed system to deliver pressure to the lungs. ENT UK recommend the strategies to minimize ventilator circuit disconnection and aerosol risks to staff include use of a cuffed non-fenestrated tube, use of in-line suction, and avoidance of unnecessary airway interventions.
Humidification and disposable inner cannula are common strategies to safeguard against tube occlusion from respiratory secretions and reduce suctioning requirements. If an inner cannula is used, we suggest reducing the frequency of changing, and reviewing the situation daily. ENT recommend a simple HME filter, which provides adequate humidification and does not generate aerosols. Using face masks and tracheostomy shields to mitigate the risks from aerosols.
The trachea is made of fibers, muscles and cartilages and can thus resist to the various pressures applied by the breathing mechanism and depress itself during the inspiratory collapse. Its mucous membrane is made of a pseudo-stratified ciliated respiratory epithelium with goblet cells that are responsible for muco-ciliary activity and for drainage of glandular secretions towards the pharynx. The presence of bronchial-associated lymphoid tissue (BALT) ensures cellular or humoral mucosal immunity.6 The coronavirus enters cells by binding to ACE2 receptor which is found on the apical surface of differentiated ciliated respiratory epithelia. This cell type is particularly dense in airway epithelial cells, hence the severity of COVID-19 disease in lungs and distal airways.7 Bronchoscopy finding were extensive bronchial mucosal hyperemia, swelling, mucus-like secretions in the lumen and jelly-like sputum blocking the airway in COVID-19 patients.8
Flexible bronchoscopy is versatile, easy to use and well tolerated in mechanically ventilated COVID-19 patients. Its applications include: (1) Collection of respiratory specimens from the lower respiratory tract (i.e. sputum, endotracheal aspirate, bronchoalveolar lavage) for SARS-CoV-2 or other pathogens guides the selection of appropriate antimicrobials, which may lead to clinical benefits. (2) Can be used for localization of the site of bleeding, cessation of hemoptysis, sputum or blood clots removal; if the site of bleeding is identified by bronchoscopy, local injection of cold saline, epinephrine, vasopressin, or fibrin as well as laser treatment can be performed via the bronchoscope. (3) Assist in the establishment of artificial airways; guide tracheal intubation or percutaneous tracheotomy. (4) Drugs such as infusion of α-interferon and N-acetylcysteine can be administrated via the bronchoscope.8
CONCLUSION
SARS-CoV‑2 infection may lead to acute myocardial injury through viral systemic inflammation and the interaction mechanism between SARS-CoV‑2 and ACE2, although specific mechanism remained uncertain. A thick mucus plaque and stool cell may be a specific clinical features in COVID-19 patients.
Tracheostomy has a continuing role in managing weaning from extended periods of mechanical ventilation during the COVID-19 pandemic, but the procedure might not always provide benefit, and tracheostomy and subsequent care pose risks to health-care workers. In the Age of COVID-19 pandemic, taking into account a range of considerations to do the tracheostomy, including potential risks and benefits for the individual patient; risks posed to health-care workers, other patients, and family; and available health-care resources.
KEYWORDS
SARS-CoV-2, COVID-19, Cardiovascular, STEMI
REFERENCES
1. The European Society for Cardiology. ESC Guidance for the Diagnosis and Management of CV Disease during the COVID-19 Pandemic. https://www.escardio.org/Education/COVID-19-and-Cardiology/ESC-COVID-19-Guidance. (Last update: 28 May 2020). Accessed 10 June 2020.
2. Basu-Ray I, Soos MP. Cardiac Manifestations Of Coronavirus (COVID-19) [Updated 2020 Apr 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan. Pubmed
3. Alhogbani, T. Acute myocarditis associated with novel Middle East respiratory syndrome coronavirus. Ann. Saudi Med. 36, 78–80 (2016). CrossRef Pubmed
4. Xiong TY, Redwood S, Prendergast B, Chen M. Coronaviruses and the cardiovascular system: acute and long-term implications. Eur. Heart J. 2020 Mar 18. CrossRef Pubmed
5. Mao Y, Lin W, Wen J, Chen G. Clinical and pathological characteristics of 2019 novel coronavirus disease (COVID-19): a systematic reviews (Version 2). MedRvix. 2020 Mar 6. CrossRef
6. ENT UK. Tracheostomy guidance during the COVID-19 Pandemic. https://www.entuk.org/tracheostomy-guidance-during-covid-19-pandemic. Accessed 11 July 2020.
7. M. Hitier, M. Loälec, V. Patron, E. Edy, S. Moreau[Trachée: anatomie, physiologie, endoscopie et imagerie]. EMC Oto-rhino-laryngologie, 8 (2013), pp. 1-18.
8. Liang T. Handbook of COVID-19 Prevention and Treatment. China: Jack Ma Foundation. March 2020.
Copyright Information
